Vyntus ECG
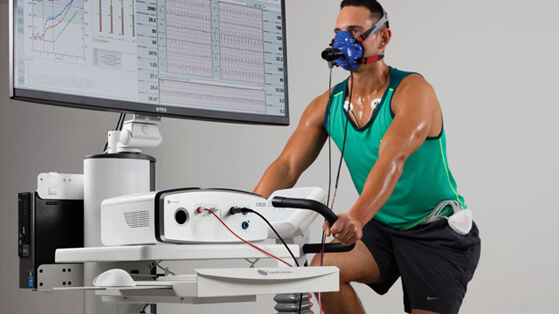
Wireless ECG in ergospirometry: Support from the initial concept right up to approval of the medical system.
On behalf of CareFusion Germany 234 GmbH (Höchberg, Deutschland), a subsidiary of Becton, Dickinson and Company, Corscience recently helped design a system for the wireless documentation, analysis and interpretation of ECG signals. The individual components have been granted national and international approvals such as the CE declaration of conformity and FDA 510 (k) so as to allow the system to be used in ergospirometry around the world.
The part of the system developed by Corscience comprises a 12-lead ECG amplifier with electrode contact and pacemaker detection as well as software for Windows® PCs. A Bluetooth® connection ensures rapid, wireless data transfer between the amplifier and PC. The device can be run on standard or rechargeable batteries, thus guaranteeing the attending physician the highest possible degree of flexibility. Compliance with the ECG standard IEC 60601-2-25 was verified in Corscience’s in-house testing laboratory in cooperation with an accredited testing institute. Conformity with additional important standards such as DIN EN 60601-1 and DIN EN 60601-1-2 has also been confirmed.
The PC software developed is based on the programming language C++ / MFC / DirectX and produces a 12-lead ECG display with an analysis function. Thanks to the modular design of the software components, creation and maintenance of different product versions is child’s play. Corscience’s well-known HES® library (HES® EXERCISE and HESHES® RESTING) guarantees precise diagnostic interpretation of the ECGs taking cardiological factors into consideration.
The device is manufactured at Corscience’s production facility in Erlangen, Germany, where the final assembly and inspections are performed on the customer’s behalf at the premises specifically designed to offer flexibility.

„Together with Corscience, we developed an innovative product. It is based on proven technology and has been marketed successfully. I appreciate the know-how of Corscience in the areas of ECG as well as Mobile Health. I value CS’ very constructive approach to cooperation.“